Ex Vivo Efficacy of Anti-cancer Peptide Pnc-27
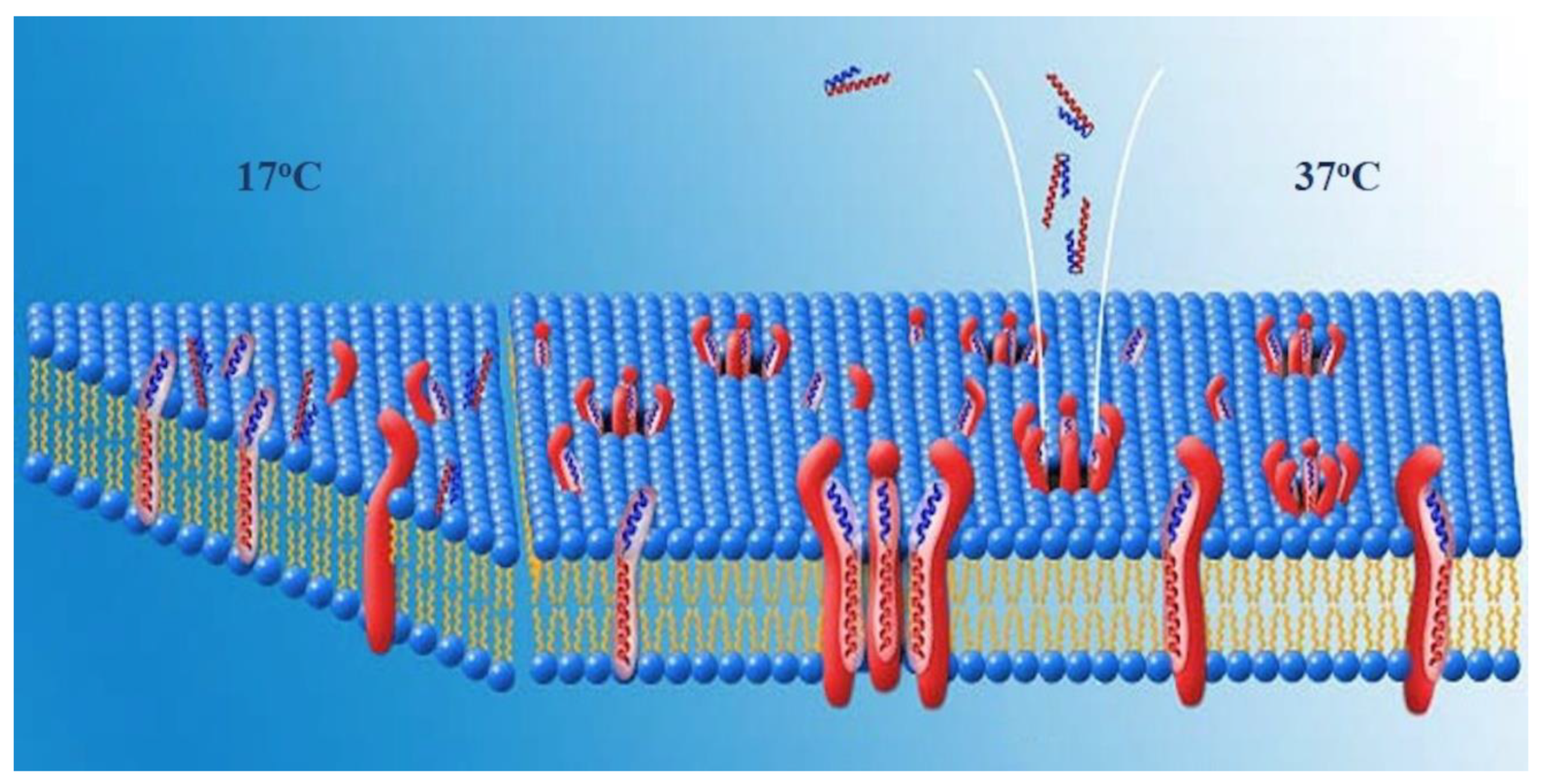
Anticancer peptide, PNC-27, is a membrane-active peptide that binds to the HDM-2 protein produced in the cancer cell membranes of tissue tumor cells. The peptide stimulates transmembrane pore development in cancer but not in normal cells, resulting in tumor cell necrosis (cell death).
PNC-27 has proved to be very successful in targeting a broad number of particular kinds of cancer, such as pancreatic cancer, breast cancer, leukemia, melanoma, and more. Buy PNC-27 peptide only if you are a licensed professional.
What is PNC-27?
Immunotherapeutic substances have been explored for many decades as an alternative therapy for cancer. Current research suggests that immunotherapy is safer than radiation or chemotherapy.
This process is linked to immunotherapy’s strategy of harnessing the body’s defensive system instead of ingesting dangerous substances. Two kinds of immunotherapy that have proved successful in healing cancer without the extra hazard of chemical toxicity are PNC-27 and PNC-28. A supercomputer generated these peptides at SUNY Downstate Medical Center in New York in 2000. PNC-27 and PNC-28 work with the MDM2 -P53 tumor suppressor complex. It is a competitive inhibitor for binding, increasing the half-life of P53 in the cell and helping the clearance of cancer cells. These immunotherapy medications also can attach to the cell membrane and lyse the cell. The clinical studies for PNC-27 and 28 were successful. Research suggests that this immunotherapy may eradicate cancer and guarantee that a recurrence does not develop.
How does PNC-27 peptide work?
HDM-2 is found in cancer cell membranes. As soon as PNC-27 is injected, it makes its way to the HDM-2 on the surface of cancer cell membranes. They puncture the cell membrane when they bind, killing the cell within. In turn, this causes the cancer cell to die.
The Anticancer Peptide PNC-27 Is Effective Against Patient-Derived Epithelial Ovarian Cancer When Administered Ex vivo.
The goal is to reduce the high mortality rate of epithelial ovarian cancer, which is the worst of all gynecologic cancers, despite an 80% response rate to chemotherapy. PNC-27 and PNC-28 are anticancer peptides that have preferentially destroyed many cancer cells in vitro and in vivo. Researchers hypothesized that PNC-27 would help combat human primary epithelial ovarian cancer; therefore, the researchers set out to do that.
Methods: Primary cultures were created using freshly obtained epithelial ovarian cancer cells from individuals with newly diagnosed ovarian cyst adenocarcinomas. Researchers isolated mucinous cystadenocarcinoma and high-grade papillary serous carcinoma cell lines. Experts studied these cells’ morphological characteristics, growth needs, and serum independence in vitro to describe their malignant qualities. To quantify the effects of PNC-27 treatment, they measured the suppression of cell growth with the MTT cell proliferation test and the direct cytotoxicity with the lactate dehydrogenase assay (LDH).
Conclusions: PNC-27 is cytotoxic and suppresses the proliferation of human primary cancer cells newly isolated from two ovarian epithelial tumors in a dose-dependent manner. The findings showed that primary cancer cells were unaffected by the presence of the control peptide PNC-29. Researchers also found that PNC-27 exhibited cytotoxicity against cells derived from well-established, chemotherapy-resistant ovarian cancer cell lines.
These results demonstrate PNC-27’s effectiveness on primary cancer cells for the first time, which is a significant step forward in the field. According to the findings, PNC-27 peptide may serve as an effective alternative therapy for newly diagnosed ovarian cancer cases and those that have developed resistance to standard chemotherapies.




